 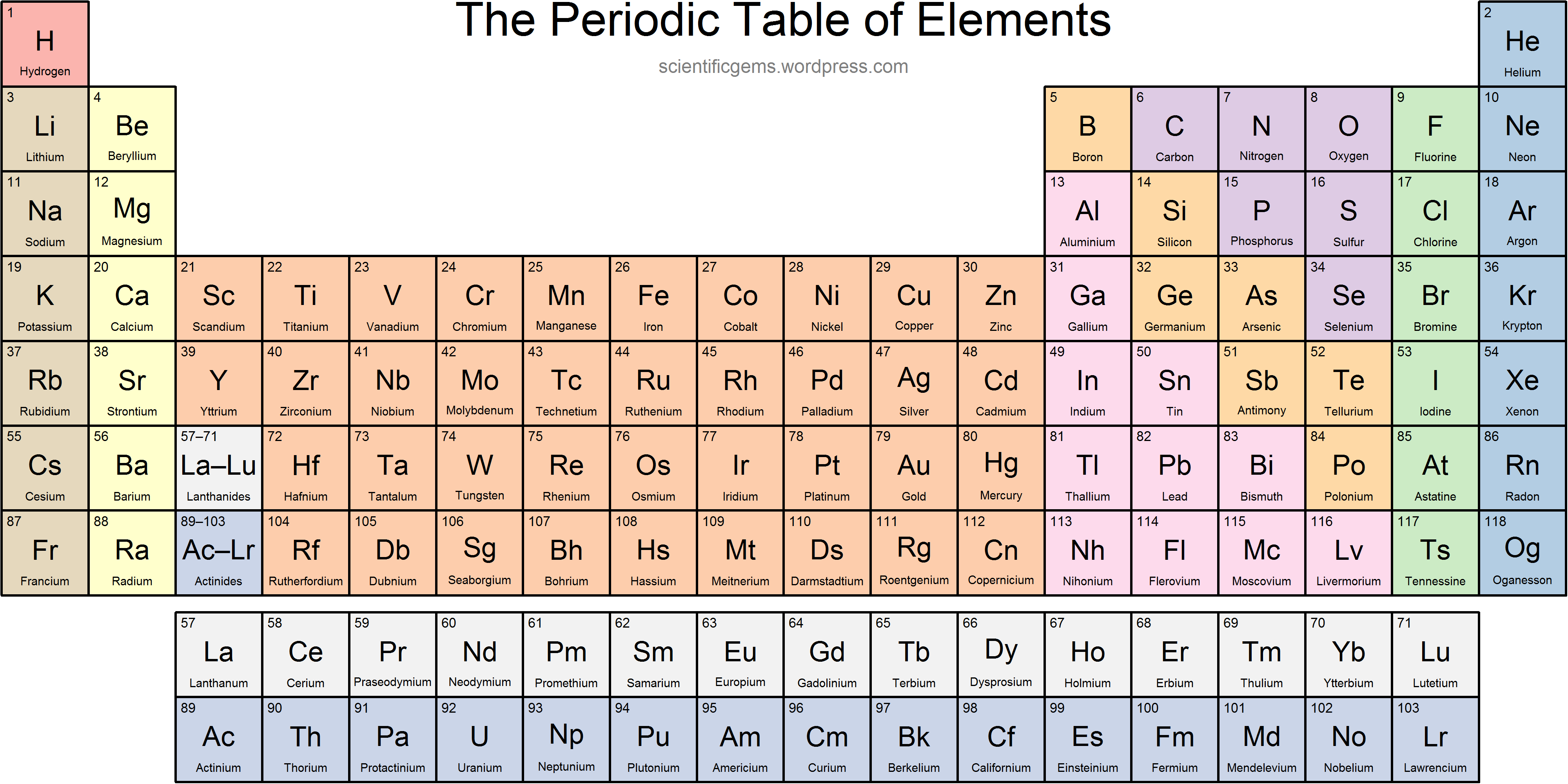
The element’s number is its atomic number, which is the number of protons in atoms.īy dynamic periodic table, you get all information about the periodic table, and here can easily cover all information regarding every element including everything like electrons, protons, neutrons, electrons on outermost orbits and temperature, etc. As you know every element consists of one or two letters symbol, which is a kind of form of present and old names. Now I provide you with all periodic table elements with names with the increasing form of an atomic number. Here you can also Download the Periodic Table with Charges of Elements. It consists of two protons and two electrons, and it is the most common form of hydrogen because it is stable with neutral charges, it is also an antioxidant in hydrogen-rich water, molecular hydrogen is the smallest molecule in the universe, which means that no one can go this molecule can easily go. 
Molecular hydrogen is the form where when two hydrogen atoms make a bond together and hydrogen molecules. it takes a minus sign and it makes a regular Hydrogen atom. The hydride is hydrogen which has an extra electron so it takes a negative charge ion or anion, i.e. Proton is a form of hydrogen that produces the ATP enzyme. When we listen to the word proton we easily assume that it means positive and it also means positive ions of hydrogen are known as a proton. In the periodic table, Hydrogen is on top and it consists of 1 proton and 1 unpaired electron which we consider a free radical, and this unpaired electron is eagerly waiting for making a pair with other electrons. Those that have more electrons are considered stable atoms. The atom of elements is stable when its outermost cell is filled with outer electrons or half-filled. As we all know charges of an atom are based on the valence electron or oxidation state. You can also use this periodic chart to know more about the charges of elements and one thing you know is how those elements make a bond with other elements. Periodic Table with Atomic ChargesĪs we all know the value of the periodic table is made by charges for atoms of the chemical elements. A student can fill and this periodic table helps them improve learning. There we see a basic table that students can also fill by their end which we call the Blank periodic table. You can also print any interactive periodic table by your end which helps you to know more about this, there are some elements that are fundamentally different from the conventional periodic table and which are defined by charge positive or negative, and valence statements were taken by these elements and these are the few things which make the periodic table much more effective.įor this, there are also lots of books on the periodic table and every book provides the same information in different ways we can’t neglect this because with every book we learn something new. Please report any accidental mistake in the above statistics on chemical elements.As we all know there are 118 elements in the periodic table and this periodic table cites keeping with most IUPAC values and by this, you can use it for your calculation and homework. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.Ĭlick here: for a schematic overview of the periodic table of elements in chart formĭo you need to know the weight of some molecules? Try our Molecular Weight Calculator! Please note that the elements do not show their natural relation towards each other as in the Periodic system. The first chemical element is Actinium and the last is Zirconium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is alphabethically listed. This list contains the 118 elements of chemistry. Separation and Concentration Purification RequestĬhemical elements alphabetically listed The elements of the periodic table sorted by name in an alphabetical list.Ĭlick on any element's name for further chemical properties, environmental data or health effects. 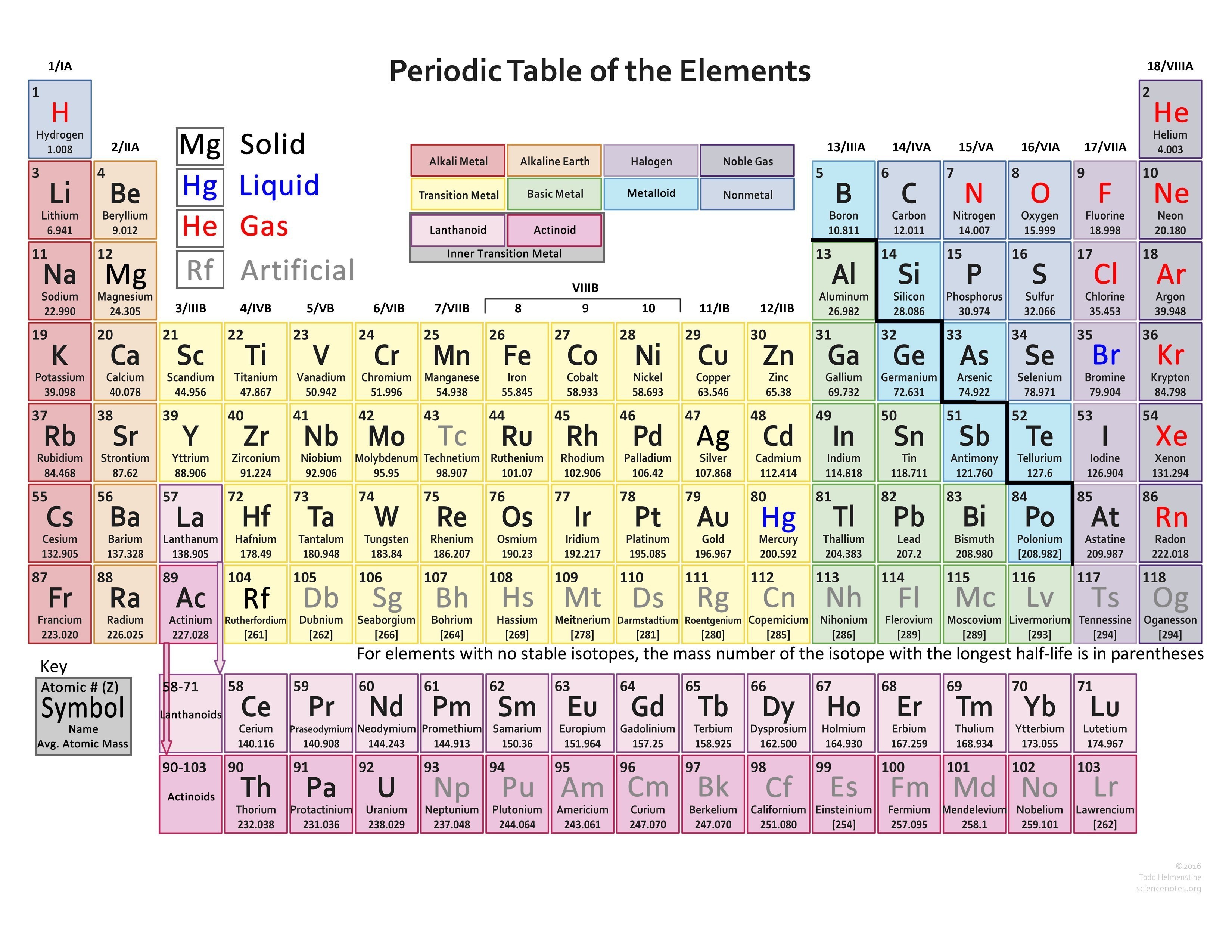
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
